Current threats to coastal seagrass ecosystems
By Hanover Matz, RJD Intern
If asked what marine ecosystems are currently most at risk, many people would probably identify coral reefs and mangrove forests. Climate change, sea level rise, and habitat degradation are all terms that come to mind when addressing the decline of corals and mangroves worldwide. However, equally important and at risk are seagrass ecosystems. Seagrasses are marine flowering plants that form ecologically important coastal habitats in tropic and temperate oceans, playing a key role in unison with coral reefs and mangroves (Short et al., 2011). These three habitats exchange nutrients and organic matter, and seagrasses provide important habitat for many species of marine fauna and juvenile fish (van Tussenbroek et al., 2014). Endangered megafauna such as manatees, dugongs, and sea turtles graze on seagrass beds. In addition to supporting marine biodiversity, seagrass beds provide many benefits to human society. They support fisheries and provide livelihoods for millions of people in coastal communities (Short et al., 2011). Seagrass beds also maintain water quality and reduce turbidity through sediment deposition. By acting as nurseries for many economically important fish species such as snapper and grouper, they help support both tourism and fisheries (Lirman et al., 2014). Due to increasing anthropogenic threats to seagrass ecosystems, we are in danger of losing these important benefits.

A green sea turtle grazes on seagrass, an important food source for this endangered species. Photo courtesy of P. Lindgren via Wikimedia Commons
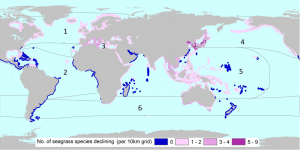
In order to implement conservation measures, the current status of seagrass species must be established. An evaluation of the world’s seagrass species by Short et al. (2011) utilized criterion set forth by the International Union for the Conservation of Nature (IUCN) to determine the risk of extinction for each seagrass species. The IUCN uses extinction risk theory and data on population reduction and geographic range to determine the conservation status of a species, ranked as Extinct, Critically Endangered, Endangered, Vulnerable, Near Threatened, Least Concern or Data Deficient. Seagrass experts used these criteria along with data on 72 species of seagrass to determine the vulnerability of each species. Based on the results of Short et al., fifteen species of seagrass were found to be threatened (Endangered/Vulnerable) or Near Threatened, with three considered Endangered. While forty-eight seagrass species were considered Least Concern, twenty-two were considered to have declining populations. This evaluation shows that while only a few seagrass species may be currently threatened with extinction, if population trends continue, many more species may be facing significant reductions in geographic range in the future. Figure 2 below shows the number of seagrass species with declining populations across the globe. The region with the highest number of declining species, the coasts of China, Japan, and Korea, corresponds with areas of high human development.
Along with the IUCN Red List evaluation, the Caribbean Coastal Marine Productivity (CARICOMP) program has monitored seagrass communities in the Caribbean from 1992-2007 for changes in biomass and productivity. With data taken from 52 monitoring stations across the Caribbean, van Tussenbroek et al. (2014) assessed the impact of human activities on seagrass habitats. Forty-three percent of the seagrass communities at thirty-five of the long-term monitoring stations showed changes in biomass and productivity associated with environmental degradation. The authors indicated increased terrestrial run-off (sewage, fertilizer, and/or sediments) as the major anthropogenic influence on seagrasses in the Caribbean. Figure 3 shows the distribution of human impacted monitoring stations across the Caribbean. Like the IUCN Red List evaluation by Short et al., this study shows that we are just beginning to understand the effects human activities have had on seagrass habitats, and that these impacts will likely increase in the near future.
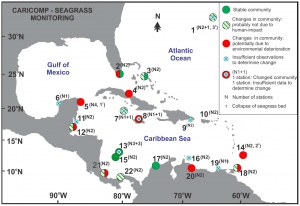
Distribution of seagrass community monitoring stations in the Caribbean, indicating communities potentially altered by environmental degradation (van Tussenbroek et al., 2014)
If seagrasses are on the decline, what is that primary cause of this decline? By combining the knowledge of several seagrass experts, Grech et al. (2012) identified many of the major threats to seagrass communities. Of the threats assessed, industrial and agricultural run-off, coastal infrastructure development, and dredging were determined to have the greatests impacts on seagrasses globally. These anthropogenic activities disturb seagrasses by increasing water turbidity and physically damaging seagrass habitat. Aquaculture development, trawling, and boat damage can also harm coastal seagrass communities. In addition to these direct human activities, climate change, sea level rise, and increasing severity of tropical storms were seen as potential risks for seagrasses (Grech et al., 2012). An example of how human activity can alter seagrass communities in South Florida was demonstrated by a study conducted in western Biscayne Bay (Lirman et al., 2014). Lirman et al. found that the proximity of the major metropolitan center, Miami, and changes in hydrology due to efforts to restore the Everglades have caused shifts in coastal salinity. These changes in turn have altered the composition of seagrass communities composed primarily of Thalassia, Halodule, and Syringodium seagrass species. Changes in salinity and nutrient availability may possibly cause the decline of seagrass dominated communities in exchange for macroalgae domianted communities. Once again, the association of human development can have serious consequences for coastal ecosystems.
In the face of all of these threats, what can be done to protect and conserve seagrasses? In an evaluation of coastal resource degradation, Wilkinson and Salvat (2012) assessed possible management solutions to help protect coral reefs, mangroves, and seagrasses. These resourcees have often been described as “commons”, open for access to anyone, but in reality these resources generally fall under the control of local coastal communities. In order to manage seagrasses, effective policies must be implemented at the local level. However, there is a disconnect between the regions of conservation research (developed nations), and the primary regions of seagrass habitat (developing nations). If seagrasses are to be protected through the use of management and Marine Protected Areas (MPAs), there must be greater cooperation between governments, policy makers, and scientists both at the national and international level (Wilkinson and Salvat, 2012). The global status of seagrass species and the current threats facing them have been established. While more research will certainly be beneficial, we need to focus on reducing the impacts of human activities. The best possible management effort will take into account all users of seagrass ecosystems, so that they can be used but not overexploited for future generations.
References
- Grech, A., K. Chartrand-Miller, P. Erftemeijer, M. Fonseca, L. McKenzie, M. Rasheed, H. Taylor and R. Coles (2012). “A comparison of threats, vulnerabilities and management approaches in global seagrass bioregions.” Environmental Research Letters 7(2): 024006.
- Lirman, D., T. Thyberg, R. Santos, S. Schopmeyer, C. Drury, L. Collado-Vides, S. Bellmund and J. Serafy (2014). “SAV Communities of Western Biscayne Bay, Miami, Florida, USA: Human and Natural Drivers of Seagrass and Macroalgae Abundance and Distribution Along a Continuous Shoreline.” Estuaries and Coasts 37(5): 1243-1255.
- Short, F. T., B. Polidoro, S. R. Livingstone, K. E. Carpenter, S. Bandeira, J. S. Bujang, H. P. Calumpong, T. J. B. Carruthers, R. G. Coles, W. C. Dennison, P. L. A. Erftemeijer, M. D. Fortes, A. S. Freeman, T. G. Jagtap, A. H. M. Kamal, G. A. Kendrick, W. Judson Kenworthy, Y. A. La Nafie, I. M. Nasution, R. J. Orth, A. Prathep, J. C. Sanciangco, B. v. Tussenbroek, S. G. Vergara, M. Waycott and J. C. Zieman (2011). “Extinction risk assessment of the world’s seagrass species.” Biological Conservation.
- van Tussenbroek, B. I., J. Cortes, R. Collin, A. C. Fonseca, P. M. Gayle, H. M. Guzman, G. E. Jacome, R. Juman, K. H. Koltes, H. A. Oxenford, A. Rodriguez-Ramirez, J. Samper-Villarreal, S. R. Smith, J. J. Tschirky and E. Weil (2014). “Caribbean-wide, long-term study of seagrass beds reveals local variations, shifts in community structure and occasional collapse.” PLoS One 9(3): e90600.
- Wilkinson, C. and B. Salvat (2012). “Coastal resource degradation in the tropics: does the tragedy of the commons apply for coral reefs, mangrove forests and seagrass beds.” Mar Pollut Bull 64(6): 1096-1105.