Coral Reefs and the Threat of Ocean Acidification
by Hanover Matz, RJD Intern
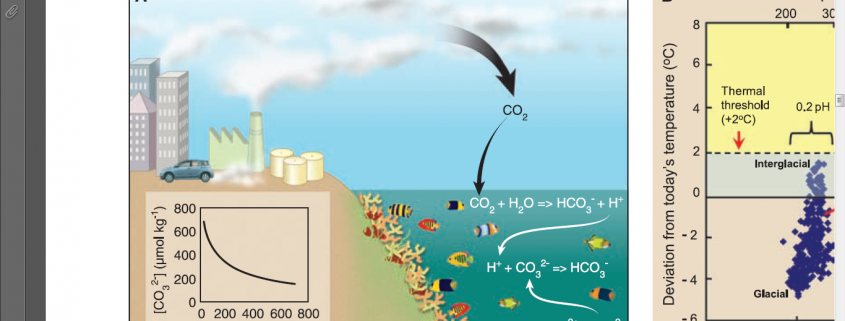
While global climate change is often the environmental concern at the forefront of the discussion about greenhouse gas emissions, ocean acidification is a marine conservation issue just as closely tied to the amount of carbon dioxide (CO2) humans have put into the atmosphere since the Industrial Revolution. It is understood that the oceans act as a sink for atmospheric CO2: as humans increase the amount of carbon dioxide in the atmosphere by burning fossil fuels, more carbon dioxide diffuses from the atmosphere into the world’s oceans. This increase in the uptake of CO2 affects the ocean by reducing the pH, or increasing the acidity, of seawater, an effect known as ocean acidification (Kleypas et al. 2006). Chemically, ocean acidification occurs through the following process: an increase in the concentration of CO2 in the water leads to an increase in the concentration of two chemicals: bicarbonate (HCO3–) and hydrogen ions (H+). By increasing the concentration of H+, the pH of the water is lowered and becomes more acidic. This shift in equilibrium towards bicarbonate and hydrogen ions also causes a shift in the chemistry of calcium (Ca2+) and carbonate (CO32-) ions. Hydrogen ions react with available carbonate ions to produce more bicarbonate, a process which reduces the formation of solid calcium carbonate (CaCO3). Thus ocean acidification has two significant chemical effects on the marine environment: it lowers the pH and decreases the availability of carbonate (Hoegh-Guldberg et al. 2007)
What does this mean for coral reefs? The hard coral species that make up reefs today belong to the order Scleractinia. These scleractinian corals are a colony of polyps that form a hard exoskeleton by secreting aragonite, a solid form of calcium carbonate. Increasing ocean acidification reduces the availability of carbonate in the water as well as the pH, so it is more difficult for the corals to form necessary hard skeletons. Many cellular and physiological responses have been observed in corals subjected to increased acidification, as shown in a 2012 study by Kaniewska et al. on Acropora millepora. The corals in the study were subjected to increasing levels of CO2, and were shown to exhibit changes in metabolism, calcification, and cellular activity. Not only do high levels of CO2 make it more difficult for corals to calcify, or form hard skeletons, due to the lack of carbonate, but they make the energy investment in calcification for the coral more costly. Corals rely on endosymbiotic algae in their cells known as Symbiodinium, or zooxanthellae, for energy from photosynthesis. Kaniewska et al. showed that increasing the level of CO2 caused the coral branches to lose their symbiotic algae, a process normally caused by increasing ocean temperature known as bleaching. Those corals that retained their zooxanthellae exhibited a 60% reduction in net photosynthesis per cell. A reduction in photosynthesis means less available energy to coral polyps, which in turn reduces coral health and reproductive ability. The study also indicated an increase in internal cellular pH regulation by the corals due to changes in CO2 levels. Increasing internal pH regulation may result in less energy being devoted to calcification. By decreasing calcification, not only does ocean acidification decrease coral growth, but it also decreases the accretion of the reef system as a whole.
Why do these physiological effects on corals matter to the reef ecosystem, or to human society? Corals constitute the primary three dimensional structures of most reef systems; any negative effect to their health will detrimentally affect the health of the reef. A study by Hoegh-Guldberg et al. published in 2007 demonstrated the effect increasing ocean acidification will have on coral reef ecosystems. The use of field studies and experimental simulations produced a model that showed as global ocean temperatures rise and pH levels fall due to increasing atmospheric CO2, it is expected that coral dominated communities will be replaced by macroalgae and non-coral dominated communities. The basic cause behind this is decreased coral calcification: if it becomes harder for the corals to produce their calcium carbonate skeletons, their structures will become weaker, their growth decreases, they may be eroded or damaged, and they will be outcompeted by other species, specifically macroalgae. The stress induced by ocean acidification may also cause reduced coral reproduction, yet another factor leading to decreased coral dominated reefs. Without corals, the biodiversity of a reef system greatly decreases as there is no longer a viable habitat for many fish species. For humans, this means significant potential damage to both fishing and tourism industries that rely on coral reefs and the fish they support. Without tourism and fishing, many countries would not only lose a significant source of income, but a significant food source for their growing populations. Coral reefs also provide protection from wave action and storms, reducing coastal erosion. The study indicates that the model takes into account atmospheric CO2 increases at the lower end of predictions for the coming century. The authors astutely note that it is “sobering” to realize these serious effects on coral reefs are based on the most optimistic outcomes of atmospheric CO2 and global temperature changes.

Potential dominant reef communities at predicted levels of atmospheric CO2 and ocean temperature increases (Hoegh-Guldberg et al. 2007)
Is there any hope for coral reefs? Is it at all possible that they can adapt to the threat of ocean acidification? One study does indicate that some corals may have the ability to adjust to decreasing ocean pH. McCulloch et al. published a study in 2012 that focused on the ability of corals to up-regulate their internal pH levels. Corals precipitate new calcium carbonate in a fluid between the existing skeleton and part of the polyp known as the calicoblastic ectoderm. At this calicoblastic layer, corals are capable of increasing the pH relative to the pH of ambient seawater in order to facilitate calcification. The study results indicate that for some coral species, as the ambient seawater pH decreases due to acidification, the corals are capable of further up-regulating their internal pH in response in order to reduce the overall internal change in pH and to continue to calcify. This up-regulation of internal pH results in higher coral calcification rates compared to abiotic or chemical precipitation of calcium carbonate at the same seawater pH. The coral species demonstrate an ability to adjust their internal pH in order to continue calcifying in acidic conditions. Does this mean these coral species will be better able to survive increasing ocean acidification? Perhaps, but the study indicates that it is necessary for the corals to maintain their symbiotic relationship with zooxanthellae in order to produce the energy needed for calcification. The loss of zooxanthellae to stress or bleaching events would reduce the effectiveness of this ability. While some corals may exhibit less sensitivity to pH changes than others based on their ability to up-regulate internal pH, all coral species will likely have difficulty adapting to not only ocean acidification, but the combined effects of ocean acidification, changes in ocean temperature, and the impact of human pollution.

Seawater pH versus internal pH of calcifying fluid of coral species. Foraminifera (forams), another type of calcifying marine organism, do not exhibit this ability to up-regulate internal pH (McCulloch et al. 2012)
Ocean acidification is a significant threat to the health of coral reef systems. What can be done to prevent potential damage from acidification? In the face of this danger to reef ecosystems, there are possible conservation methods that can be taken to protect coral species. Coral reefs that are already in a healthy state are better prepared to handle changes in pH than those suffering from other environmental stressors. Reefs with stable levels of herbivorous grazers, such parrotfish or the sea urchin Diadema antillarum, are also more resilient to stress due to reduced competition with algae (Hoegh-Guldberg et al. 2007). Effective conservation management of coral reefs provides the best method for ensuring their survival. Continuing research to determine how to mitigate the effects of acidification is also necessary. As coral reefs are threatened by climate change, pollution, and other human induced stressors, ocean acidification will remain one serious part of the ongoing endeavor to protect coral reefs.
REFERENCES
- Hoegh-Guldberg, Ove, et al. “Coral reefs under rapid climate change and ocean acidification.” Science 318.5857 (2007): 1737-1742.
- Kaniewska, Paulina, et al. “Major cellular and physiological impacts of ocean acidification on a reef building coral.” PLOS ONE 7.4 (2012): e34659.
- Kleypas, J.A., R.A. Feely, V.J. Fabry, C. Langdon, C.L. Sabine, and L.L. Robbins, 2006. Impacts of Ocean Acidification on Coral Reefs and Other Marine Calcifiers: A Guide for Future Research, report of a workshop held 18–20 April 2005, St. Petersburg, FL, sponsored by NSF, NOAA, and the U.S. Geological Survey, 88 pp.
- McCulloch, Malcolm, et al. “Coral resilience to ocean acidification and global warming through pH up-regulation.” Nature Climate Change 2.8 (2012): 623-627.




Great article that summarizes the crux of ocean acidification and its effects on corals. This is really helpful for my teaching. I find making the connection between changing water chemistry an the ability of corals to calcify a difficult one for students to grasp. This will help with my explanations.